Vertiflex™ Procedure for Lumbar Spinal Stenosis (LSS)
Now Offering: The Vertiflex™ Procedure for Lumbar Spinal Stenosis (LSS)
We are excited to announce that Dr. Lance Larsen and Dr. Mark Sison now offer Boston Scientific's Vertiflex Procedure to our patients with lumbar spinal stenosis (LSS).
What is Lumbar Spinal Stenosis?
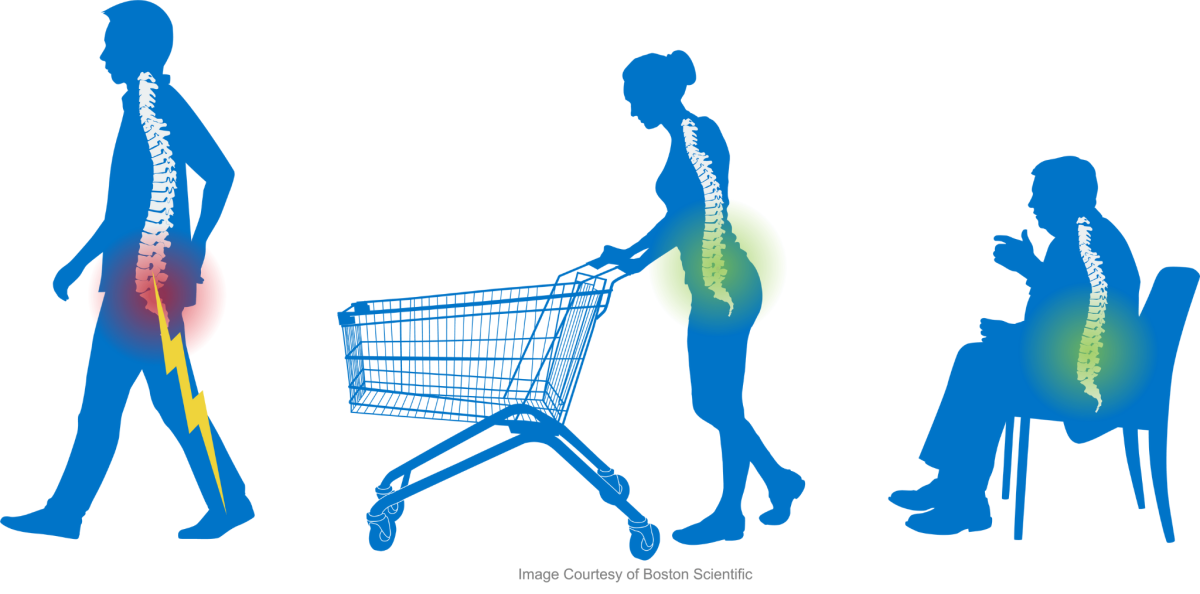
Lumbar spinal stenosis (LSS) is a gradual narrowing of the space in the spine where nerves pass through. The narrowing of the space can cause a “pinching” on the nerves in the lower back, which can result in aching, dull back pain down the buttocks and into one or both legs. This natural, degenerative condition may be the result of aging and “wear and tear” on the spine from everyday activities. The result is pain, heaviness, numbness, tingling and/or weakness in the legs, buttocks, and back.
Lumbar spinal stenosis (LSS) occurs as people age and their spines begin to wear. The result is a narrowing of space in the lower spine where nerves pass through. The narrowing of the space in the lower (or lumbar) spine is called lumbar spinal stenosis and may cause a “pinching” on the nerves in the lower back. This can result in an aching, dull back pain down the buttocks and into the legs.
Lumbar spinal stenosis may be the result of aging and “wear and tear” on the lower spine from everyday activities. Common symptoms can include pain, heaviness, numbness, tingling and/or weakness in one or both legs, buttocks, and/or back. Pain is usually worse when standing or walking. Relief is achieved by sitting down or bending/leaning forward. Lumbar spinal stenosis affects more than 14 million Americans1,2 and is most commonly found in adults age 60 or older. If left untreated, this condition can worsen over time, significantly restricting a person’s mobility and quality of life.3
What are the Signs of Lumbar Spinal Stenosis?
- Pain while walking
- Numbness or a “tingling” feeling in the legs, calves, or buttocks
- Weakness and/or loss of balance
- Aching, dull back pain spreading to the legs
- Decreased endurance during physical activities
- Pain relief is experienced when leaning/bending forward or sitting down
How does the Vertiflex Procedure work?
The Vertiflex Procedure is a safe, minimally invasive treatment that has been clinically proven to provide effective long-term relief from the pain associated with LSS.
How does the Vertiflex Procedure fit my pain?
The Vertiflex Procedure is intended to provide the same relief you experience when leaning forward or sitting down. It fits your pain by reducing or eliminating the pressure on the nerves caused by LSS. The spacer is also available in several different sizes to fit your anatomy.
Is the Vertiflex Procedure safe?
The Vertiflex Procedure was developed with patient safety and comfort in mind. Its minimally invasive approach is preferred among many doctors across the United States. General anesthesia is not required and there is minimal recovery time, getting you home the same day. The spacer is designed to support your existing anatomy and does not require removal of bone or tissue, making it a safe option for LSS treatment.
Is the Vertiflex Procedure FDA approved?
The minimally invasive Vertiflex Procedure is FDA approved and backed by a long-term clinical study.
Does the Vertiflex Procedure reduce opioid use?
In the clinical study, most patients on opioids stopped using them after the Vertiflex Procedure.4,*
How fast is the recovery?
The Vertiflex Procedure can be completed at an outpatient center so you can go home the same day of the procedure—we recommend having someone accompany you after the procedure. General anesthesia is not required. Your treating doctor will prescribe your post procedure care instructions. Most patients who undergo the Vertiflex Procedure can expect the guidelines below.
Is the Vertiflex Procedure effective?
The Vertiflex Procedure was the subject of an extremely thorough FDA clinical trial for LSS. Results from the trial proved the effectiveness of the procedure, with significant improvement in pain relief. The procedure can be completed in an outpatient setting and most patients can return home within the same day.
1. VertiFlex™ Superion™ Interspinous Spacer: PMA No. P140004.
https://www.vertiflex.com/wp-content/uploads/2016/09/Superion-PMA-Summa…. Accessed January 17, 2019.
2. Data on file. Navigant, LLC Market Sizing. October 2018.
3. Lumbar Spinal Stenosis. American Association of Neurological Surgeons.
https://www.aans.org/Patients/Neurosurgical-Conditions-and-Treatments/L…. Accessed January 17, 2019.
References: 1. Nunley PD, Deer TR, Benyamin RM, Staats PS, Block JE. Interspinous process decompression is associated with a reduction in opioid analgesia in patients with lumbar spinal stenosis. Pain Res. 2018;11:2943-2948. 2. Nunley PD, Patel VV, Orndorff DG, Lavelle WF, Block JE, Geisler FH. Five-year durability of stand-alone interspinous process decompression for lumbar spinal stenosis. Clin Interv Aging. 2017;12:1409-1417. N=88. *Decrease in the proportion of patients using opioids compared to baseline at five years.